Abstract
Introduction
Autoimmune cytopenias(AICs) are the most common autoimmune phenomenon encountered following haematopoietic stem cell transplant(HSCT) (Holbro et al. 2012). Mechanisms of autoimmunity post-HSCT may relate to genetic susceptibility(Consolini et al. 2016), failure of central and peripheral immune tolerance(King et al. 2004), and factors such as drugs and inflammation(Lang et al. 2005). The aim of this EBMT study is to describe the incidence, risk factors, diagnosis and treatment of AIC post alloHSCT for acquired aplastic anaemia (aAA), itself a recognized autoimmune disease in most patients.
Methods
EBMT centres performing alloHSCT for aAA were invited to participate in the study. Participating centres identified paediatric and adult patients with aAA treated between January 2002 and December 2012 with first alloHSCT, and confirmed whether AIC was diagnosed post-HSCT. Patients diagnosed with AIC prior to HSCT were excluded. Data for patients without AIC were extracted from the EBMT registry. Centres identifying cases of AIC provided additional data on diagnostic criteria and investigations, therapy administered and treatment outcome.
Results
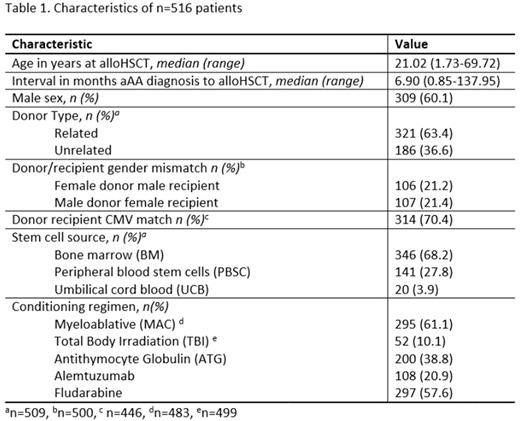
196 (38%) paediatric and 320 (62%) adult patients were confirmed eligible for study inclusion by 40 participating EBMT centres. Characteristics of n=516 patients are shown in Table 1. 23 patients were diagnosed at a median of 13.0 (range 2.6-91.4) months post alloHSCT with AICs as follows: 30.4% (n=7) immune thrombocytopenia (ITP), 26.1% (n=6) autoimmune haemolytic anaemia(AIHA), 17.4% (n=4) autoimmune immune neutropenia (AIN), and 21.7% (n=5) Evans Syndrome. 1 case (4.3%) was diagnosed both with AIHA and AIN. Cumulative incidence of AIC at 1, 3 and 5 years post AIC was 2.0% (95% CI 0.7-3.2%), 4.1% (2.3-5.9%) and 4.4% (2.6-6.3%) respectively. Cumulative incidence of AIC at 5 years post HSCT did not vary with patient age group (<18 vs ≥18), Interval from aAA diagnosis to transplant (<12 vs ≥12 months), donor/recipient CMV match, donor/recipient gender match, or conditioning with TBI (P>0.5 in all cases). However, 5-year cumulative incidence of AIC was higher amongst patients treated with non-myeloablative conditioning (6.9% vs 2.5%, p=0.03), and conditioning regimens containing Fludarabine (6.2 vs 1.9%, p=0.03) or Alemtuzumab (10.0% vs 2.93%, p=0.003). Conditioning with anti-thymocyte globulin(ATG) was associated with a lower incidence of AIC (1.6 vs 6.2%, p=0.02). Incidence of AIC was also significantly higher in patients receiving PBSC (9.9%) compared with UCB (5.0%) and BM (0.8%) (p=0.002). 2 AIC patients received a second HSCT, one 2.3 months after AIC diagnosis and one 35 months after AIC diagnosis. There were 3 deaths in the AIC group, 2 infection-related to AIC and 1 HSCT-related.
Diagnostic investigations varied across centres with direct antiglobulin test (DAT) performed in 6/7 AIHA patients, and bone marrow examination (BME) performed in 5/7 ITP patients. Anti-neutrophil antibodies were screened for in all AIN cases and detected in 1 patient. Multiple lines of therapy were required for patients with ITP, AIHA and Evans syndrome with 42.1%(n=8) of these patients not responding, and a further 26.3% (n=5) relapsing after initial complete response (CR) to first-line therapy. 1 ITP patient exhibited disease refractory to 3 lines of therapy including splenectomy. 2 AIHA patients achieved CR, and 1 patient with Evans syndrome achieved partial response following 4 lines of therapy. 2 AIN patients did not require treatment; 1 AIN patient achieved PR after first line treatment with granulocyte-colony stimulating factor (G-CSF), and a second achieved CR after second-line treatment with G-CSF and Rituximab.
Conclusions
With a cumulative incidence of 4.4% (2.6-6.3%) over 5 years, AICs are a clinically challenging and potentially life-threatening complication of alloHSCT for aAA. Patients receiving non-myeloablative conditioning regimens incorporating Fludarabine and/or Alemtuzumab, and those receiving PBSC are at higher risk of this complication. Diagnosis of AIC should be considered and investigated in cases of new or worsening cytopenia at any stage post alloHSCT for aAA. Standardization of diagnostic and therapeutic approaches, along with data registration (via the EBMT and other databases), is warranted to inform harmonized recommendations for management.
Snowden: Sanofi: Honoraria. Bader: Novartis, Medac, Amgen, Riemser, Neovii: Consultancy, Honoraria, Research Funding. Peffault De Latour: Alexion Pharmaceuticals, Inc.: Consultancy, Honoraria, Research Funding; Amgen: Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal